S2076
α-2,6-Sialyltransferase from Photobacterium damsela
recombinant, expressed in E. coli BL21, ≥5 units/mg protein
동의어(들):
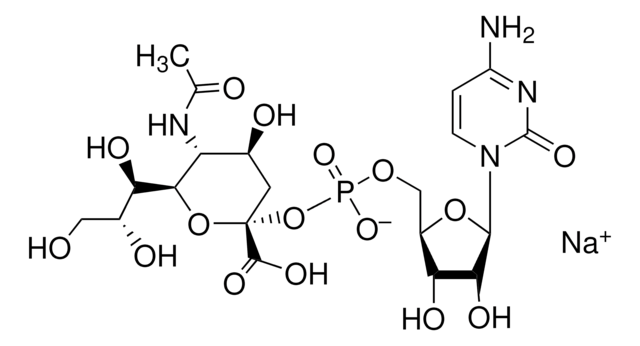
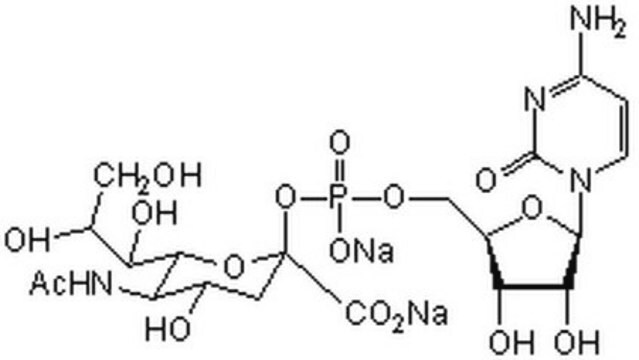
β-Galactoside α-2,6-sialyltransferase, CMP-N-Acetylneuraminate:β-D-galactosyl-1,4-N-acetyl-β-D-glucosamine α-2,6-N-acetylneuraminyltransferase
로그인조직 및 계약 가격 보기
모든 사진(2)
About This Item
추천 제품
재조합
expressed in E. coli BL21
Quality Level
양식
lyophilized powder
특이 활성도
≥5 units/mg protein
분자량
56.8 kDa
배송 상태
dry ice
저장 온도
−20°C
일반 설명
Human ST6Gal-I (β-galactoside α-2,6-sialyltransferase 1) is a member of the CAZy family GT29.
애플리케이션
α-2,6-Sialyltransferase from Photobacterium damsela has been used in resialylation and restoration of sialic acids (SAs) in HRT-18G cells.
Highly active α2-6 sialyltransferase has been used to prepare high levels of disialylated fragment crystals.
생화학적/생리학적 작용
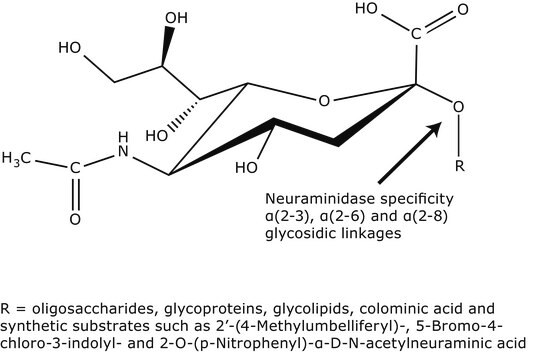
Sialyltransferase transfers Neu5Ac from CMP-Neu5Ac to the galactosyl terminus of acceptor molecules including glycoproteins, glycolipids, and oligosaccharides.
The terminal step of complex N-glycan biosynthesis is catalysed by α-2,6-sialyltransferase (STs). Bacterial α(2,6)-STs possesses broader acceptor substrate specificity when compared to eukaryotic α(2,6)-STs.
단위 정의
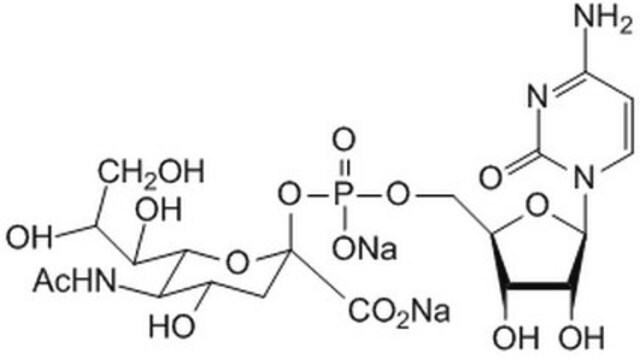
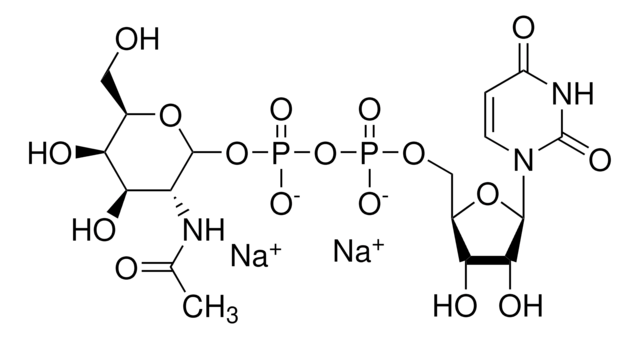
One unit will catalyze the formation of 1 μmol Neu-5-Ac-α-2,6-LacMU from CMP-Neu-5-Ac and Lac-β−OMU per minute at 37 °C at pH 8.0.
물리적 형태
Supplied as a lyophilized powder containing Tris-HCl and NaCl.
분석 메모
Enzymatic activity assays are performed in Tris-HCl buffer (100 mM, pH 8.0) containing CMP-Neu-5-Ac (1 mM) and Lac-β−OMU (1 mM) at 37 °C for 30 min and analyzed using HPLC with a fluorescence detector (excitation at 325 nm and emission at 372 nm).
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
이미 열람한 고객
High-quality production of human alpha-2, 6-sialyltransferase in Pichia pastoris requires control over N-terminal truncations by host-inherent protease activities
Ribitsch D, et al.
Microbial cell factories, 13, 138-138 (2014)
Jung-Jin Park et al.
Biochemical pharmacology, 83(7), 849-857 (2012-01-24)
β-Galactoside α2,6-sialyltransferase (ST6Gal-I) has been shown to catalyze α2,6 sialylation of N-glycan, an action that is highly correlated with colon cancer progression and metastasis. We have recently demonstrated that ST6Gal-I-induced α2,6 sialylation is critical for adhesion and migration of colon
Tatsuya Kato et al.
Journal of bioscience and bioengineering, 113(6), 694-696 (2012-02-09)
Modified polyhedrin promoter (Ppolh) was designed by repeating burst sequences (BSs) and adopted to overexpress rat α2,6-sialyltransferase (ST6Gal I) in silkworm. Modified Ppolh of five BSs with VLF-1 coexpression yielded 2.9 U/ml ST6Gal I activity and 32.5 mU/mg specific activity
Miyako Nakano et al.
Molecular & cellular proteomics : MCP, 10(11), M111-M111 (2011-08-24)
Resistance to tubulin-binding agents used in cancer is often multifactorial and can include changes in drug accumulation and modified expression of tubulin isotypes. Glycans on cell membrane proteins play important roles in many cellular processes such as recognition and apoptosis
Enhanced Bacterial alpha (2, 6)-Sialyltransferase Reaction through an Inhibition of Its Inherent Sialidase Activity by Dephosphorylation of Cytidine-5'-Monophosphate
Kang JY, et al.
PLoS ONE, 10(7), e0133739-e0133739 (2015)
문서
Glycosyltransferases were initially considered to be specific for a single glycosyl donor and acceptor, which led to the one enzyme-one linkage concept. Subsequent observations have refuted the theory of absolute enzymatic specificity by describing the transfer of analogs of some nucleoside mono- or diphosphate sugar donors.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.