추천 제품
제품명
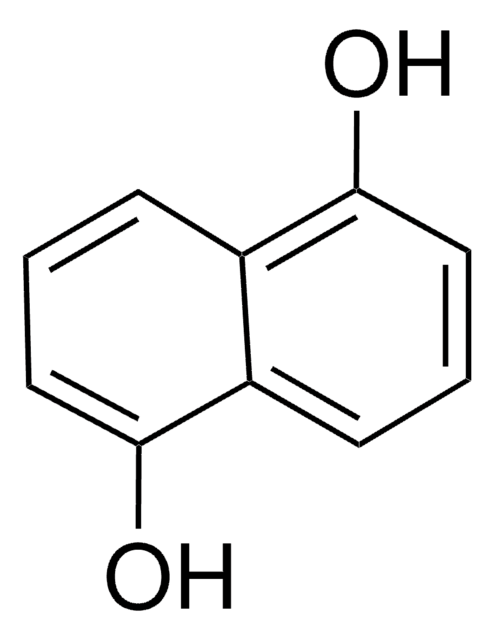
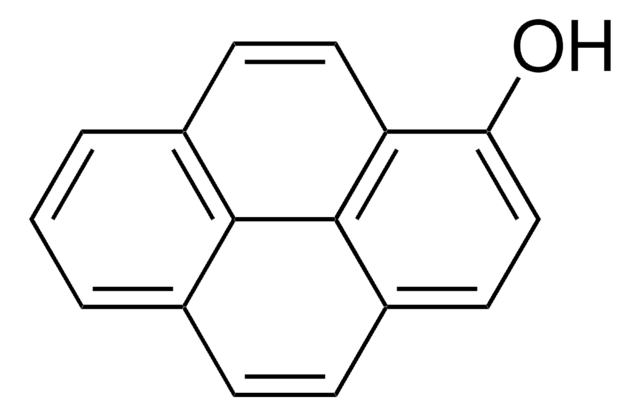
1,3-Dihydroxynaphthalene, ≥99%, crystalline
Quality Level
분석
≥99%
양식
crystalline
기술
titration: suitable
색상
white to tan
mp
123-125 °C (lit.)
124-125 °C
solubility
ethanol: 50 mg/mL
λmax
288 nm (298 nm (ethanol))
응용 분야
diagnostic assay manufacturing
hematology
histology
저장 온도
room temp
SMILES string
Oc1cc(O)c2ccccc2c1
InChI
1S/C10H8O2/c11-8-5-7-3-1-2-4-9(7)10(12)6-8/h1-6,11-12H
InChI key
XOOMNEFVDUTJPP-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
1,3-Dihydroxynaphthalene is a dye intermediate forming stable colors when applied with particular mordants and in mixtures with other oxidizing dyes. This specific reagent is intended for the quantitative colorimetric determination of alduronic acids, such as glucuronic acid from urine. The mutagenicity of ozonation derivatives of 1,3-dihydroxynaphthalene against several Salmonella typhimurium strains has been studied.
애플리케이션
1,3-Dihydroxynaphthalene is utilized in carbohydrate chromatography, sometimes in conjunction with resorcinol. It has been used incorporated into a thin-layer electrophoretic method for the analysis of biomass degradation products which contain comprising mono-, oligo- and polysaccharides, and their derivatives, in addition to phenolic compounds.
제조 메모
1,3-Dihydroxynaphthalene is soluble in ethanol (50 mg/ml), with heat as needed, yielding a clear to slightly hazy, colorless to orange-brown solution. It is also soluble in water and ether.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Muta. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
Karen A Nolan et al.
Bioorganic & medicinal chemistry letters, 20(24), 7331-7336 (2010-11-16)
The NCI chemical database has been screened using in silico docking to identify novel inhibitors of NRH:quinone oxidoreductase 2 (NQO2). Compounds identified from the screen exhibit a diverse range of scaffolds and inhibitory potencies are generally in the micromolar range.
Y Sayato et al.
Mutation research, 189(3), 217-222 (1987-11-01)
The mutagenicity of products formed by ozonation of naphthoresorcinol in aqueous solution was assayed with Salmonella typhimurium strains TA97, TA98, TA100, TA102 and TA104 in the presence and absence of S9 mix from phenobarbital- and 5,6-benzoflavone-induced rat liver. Ozonated naphthoresorcinol
Wilson Terán et al.
The Journal of biological chemistry, 281(11), 7102-7109 (2006-01-13)
The RND family transporter TtgABC and its cognate repressor TtgR from Pseudomonas putida DOT-T1E were both shown to possess multidrug recognition properties. Structurally unrelated molecules such as chloramphenicol, butyl paraben, 1,3-dihydroxynaphthalene, and several flavonoids are substrates of TtgABC and activate
Determination of glycolaldehyde in the presence of hydroxypyruvate and tartronate semialdehyde.
F E de Windt et al.
Analytical biochemistry, 106(2), 342-343 (1980-08-01)
Amy L Kieran et al.
Chemical communications (Cambridge, England), (14)(14), 1842-1844 (2005-03-30)
New macrocycles incorporating a porphyrin and a [small pi] electron-rich aromatic were prepared from a dynamic disulfide library. The outcome could be influenced by use of templates.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.