추천 제품
Quality Level
분석
98%
광학 활성
[α]20/D −116°, c = 9 in ethanol
mp
152-155 °C (lit.)
작용기
carboxylic acid
ester
phenyl
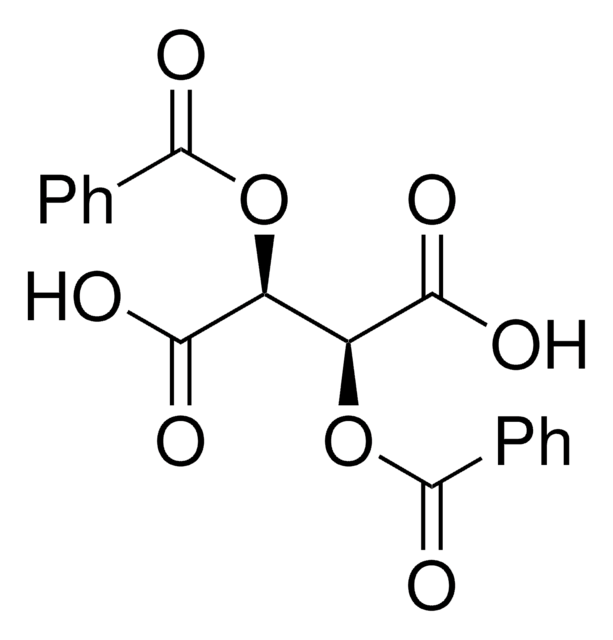
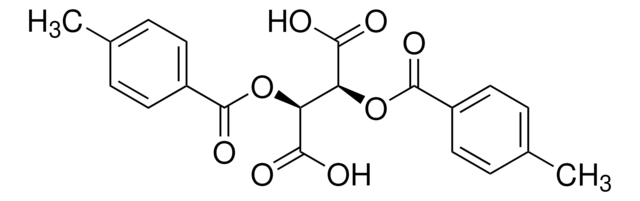
SMILES string
OC(=O)[C@H](OC(=O)c1ccccc1)[C@@H](OC(=O)c2ccccc2)C(O)=O
InChI
1S/C18H14O8/c19-15(20)13(25-17(23)11-7-3-1-4-8-11)14(16(21)22)26-18(24)12-9-5-2-6-10-12/h1-10,13-14H,(H,19,20)(H,21,22)/t13-,14-/m1/s1
InChI key
YONLFQNRGZXBBF-ZIAGYGMSSA-N
애플리케이션
Dibenzoyl-L-tartaric acid may be used as a chiral resolving agent for the resolution of racemic Troger base. It may also be used as a ligand to synthesize chiral transition metal complexes, which have potential utility in organic asymmetric catalysis.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Eye Dam. 1 - Skin Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
A convenient method for the synthesis and resolution of Troger base.
Satishkumar S and Periasamy M.
Tetrahedron Asymmetry, 17(7), 1116-1119 (2006)
Transition metal complexes of dibenzoyl-L-tartaric acid (db-L-tarH2) and L-tartaric acid (L-tarH2); X-ray crystal structure of {[Cu(L-tar)(phen)]?6H2O}n (phen=1,10-phenanthroline).
McCann M, et al.
Polyhedron, 16(20), 3655-3661 (1997)
Emily M Jutkiewicz et al.
The Journal of pharmacology and experimental therapeutics, 309(1), 173-181 (2004-01-15)
The diarylpiperazine delta-opioid agonist SNC80 [(+)-4-[(alphaR)-alpha-[(2S,5R)-2,5-dimethyl-4-(2-propenyl)-1-piperazinyl]-(3-methoxyphenyl)methyl]-N,N-diethylbenzamide] produces convulsions, antidepressant-like effects, and locomotor stimulation in rats. The present study compared the behavioral effects in Sprague-Dawley rats of SNC80 with its two derivatives, SNC86 [(+)-4-[alpha(R)-alpha-[(2S,5R)-2,5-dimethyl-4-(2-propenyl)-1-piperazinyl]-(3-hydroxyphenyl)methyl]-N,N-diethylbenzamide] and SNC162 [(+)-4-[(alphaR)-alpha-[(2S,5R)-2,5-dimethyl-4-(2-propenyl)-1-piperazinyl]-(3-phenyl)methyl]-N,N-diethylbenzamide], which differ by one
Jin-Lan Zhou et al.
Chemical communications (Cambridge, England), (48)(48), 5200-5202 (2007-12-07)
Chiral calix[4]arenes bearing long tertiary alkyl groups at the upper rim and S-1-phenylethylamine groups at the lower rim can form heat-set gels and egg-like vesicles enantioselectively with d-2,3-dibenzoyltartaric acid in cyclohexane, which is the first example of heat-set gels resulting
Christian Zimmer et al.
Applied microbiology and biotechnology, 73(1), 132-140 (2006-07-19)
In a screening procedure a pink-colored yeast was isolated from enrichment cultures with (2R,3R)-(-)-di-O-benzoyl-tartrate (benzoyl-tartrate) as the sole carbon source. The organism saar1 was identified by morphological, physiological, and 18S ribosomal DNA/internal transcribed spacer analysis as Rhodotorula mucilaginosa, a basidiomycetous
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.