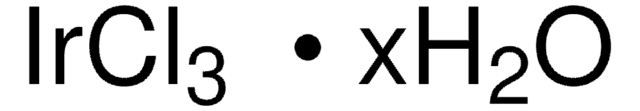377627
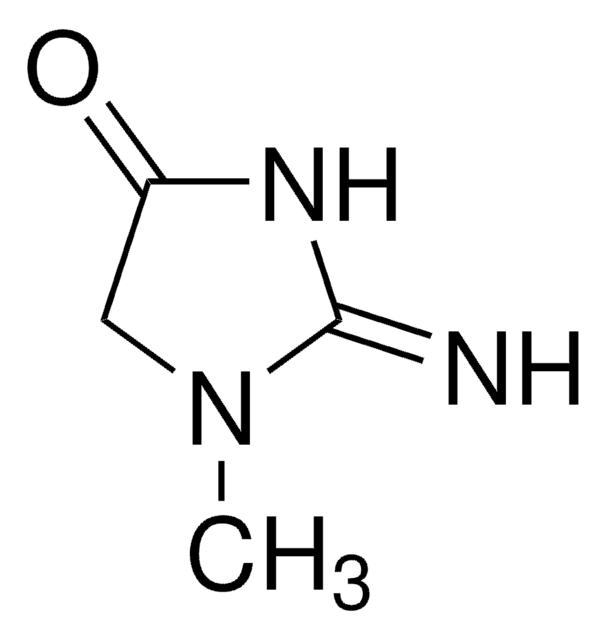
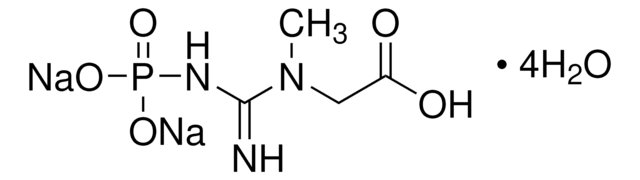
2-Imino-1-imidazolidineacetic acid
98%
Synonym(s):
1-Carboxymethyl-2-iminoimidazolidine, Cyclocreatine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C5H9N3O2
CAS Number:
Molecular Weight:
143.14
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
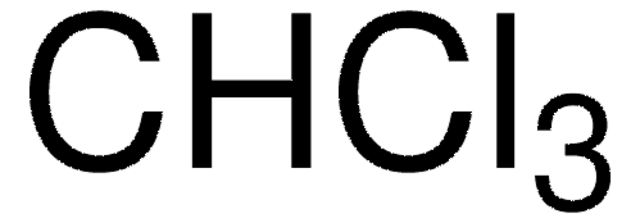
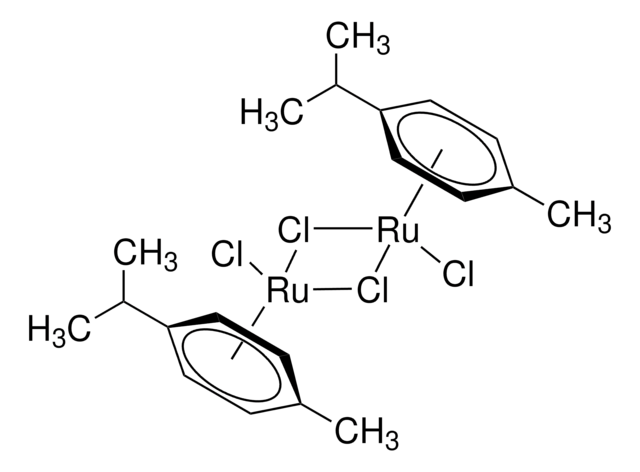
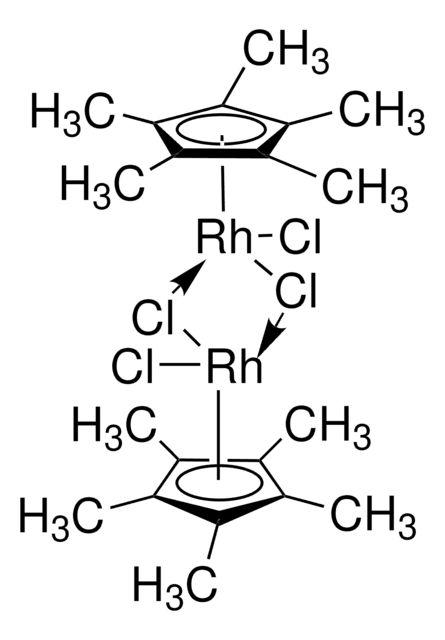
Recommended Products
assay
98%
form
solid
solubility
1 M HCl: soluble 25 mg/mL, clear, colorless
functional group
carboxylic acid
SMILES string
OC(=O)CN1CCNC1=N
InChI
1S/C5H9N3O2/c6-5-7-1-2-8(5)3-4(9)10/h1-3H2,(H2,6,7)(H,9,10)
InChI key
AMHZIUVRYRVYBA-UHFFFAOYSA-N
General description
2-Imino-1-imidazolidineacetic acid (cyclocreatine) is an analog of creatine. It is reported to exhibit antitumour effect in some transplanted human and rodent tumours in vivo. It is reported as an efficient substrate for creatine kinase. It is an anticancer and neuroprotective agent. The crystal structure of cyclocreatine has been studied by X-ray diffraction methods. It is reported to crystallize as a zwitterion in the monoclinic system.
Application
2-Imino-1-imidazolidineacetic acid (cyclocreatine) is suitable for use in a study to investigate the growth inhibitory effects of cyclocreatine on LS174T human colon adenocarcinoma implanted subdermally in nude mice. It may be used to evaluate the brain-type creatine kinase (CKB), to study the role of CKB in cigarette smoke-induced bronchial epithelial cell senescence.
Protectant against inhibition of cardiac mitochondrial respiration by methylglyoxal
Growth inhibition of Hodgkin disease-derived cell lines
Investigations into effects in rat hepatocarcinogenesis model
Growth inhibition of Hodgkin disease-derived cell lines
Investigations into effects in rat hepatocarcinogenesis model
Biochem/physiol Actions
Creatine analog that protects tissues from ischemic damage; decreases the rate of ATP production via creatine kinase and reduces the proliferation of tumor cell lines that are characterized by high levels of creatine kinase expression.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
N Askenasy et al.
The American journal of physiology, 273(2 Pt 1), C741-C746 (1997-08-01)
Creatine kinase (CK) has been implicated in affecting cell growth, and the CK substrates creatine (Cr) and cyclocreatine (CyCr) have been shown to have anti-tumor activity. The influence of Cr and CyCr on liver regeneration following major hepatectomy was evaluated
J S Cantwell et al.
Biochemistry, 40(10), 3056-3061 (2001-03-22)
Creatine kinase (CK) catalyzes the reversible phosphorylation of the guanidine substrate, creatine, by MgATP. Although several X-ray crystal structures of various isoforms of creatine kinase have been published, the detailed catalytic mechanism remains unresolved. A crystal structure of the CK
Cyclocreatine, an anticancer and neuroprotective agent. Spectroscopic, structural and theoretical study.
Pis-Diez R, et al.
Journal of Molecular Structure, 975(1), 303-309 (1995)
C B Cuono et al.
Plastic and reconstructive surgery, 101(6), 1597-1603 (1998-05-16)
A general understanding of the pivotal role of phosphocreatine (PCr) as the principal determinant of skin flap survival is now emerging. Definitive metabolic investigations using phosphorus (31P) and proton (1H) magnetic resonance spectroscopy (MRS) have established that the inability to
R W Wiseman et al.
The Journal of biological chemistry, 270(21), 12428-12438 (1995-05-26)
The hypothesis tested was whether creatine kinase (CK) equilibrates with its substrates and products in the cytosol as if in solution. We used the creatine analogs cyclocreatine (cCr) or beta-guanidopropionate (beta GPA) to test if mass action ratios (gamma) for
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service