M2824
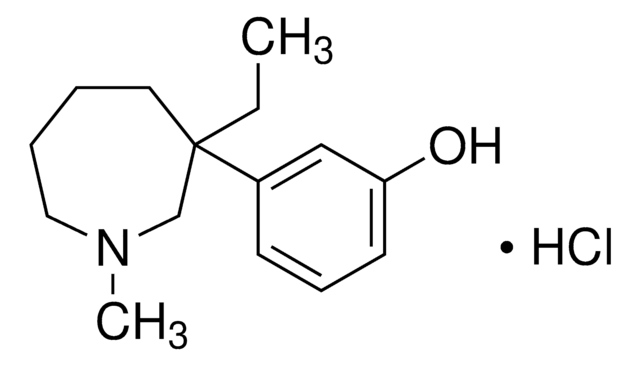
Meptazinol hydrochloride
동의어(들):
3-(3-ethylhexahydro-1-methyl-1H-azepin-3-yl)-phenol hydrochloride, IL-22811 hydrochloride, WY-22811 hydrochloride
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C15H23NO· HCl
CAS Number:
Molecular Weight:
269.81
EC Number:
MDL number:
UNSPSC 코드:
12352200
PubChem Substance ID:
NACRES:
NA.77
추천 제품
분석
≥98% (HPLC)
Quality Level
양식
powder
색상
white to off-white
solubility
H2O: >10 mg/mL
주관자
Wyeth
SMILES string
Cl.CCC1(CCCCN(C)C1)c2cccc(O)c2
InChI
1S/C15H23NO.ClH/c1-3-15(9-4-5-10-16(2)12-15)13-7-6-8-14(17)11-13;/h6-8,11,17H,3-5,9-10,12H2,1-2H3;1H
InChI key
MPJUSISYVXABBH-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
생화학적/생리학적 작용
Meptazinol entered the human pharmaceutical market as a racemic mixture in the 1980s for use as an analgesic.
Meptazinol entered the human pharmaceutical market as a racemic mixture in the 1980s for use as an analgesic. Its pharmacology is not completely understood; however, its analgesic properties are mostly due to its partial agonism at the mu1 opioid receptor. Due to its partial agonism, Meptazinol antagonizes morphine dependence in vivo. Its advantage over other opiates is its reduced capacity to cause addition and respiratory depression, also due to its intrinsic activity as a partial agonist. Meptazinol has been found to have additional activity as an acetylcholinesterase (AChE) inhibitor, particularly in its (-) enantiomeric form, which may partially explain its analgesic properties. AChE inhibitors are used to treat Alzheimer′s disease, providing additional interest in this compound.
특징 및 장점
This compound is featured on the Opioid Receptors page of the Handbook of Receptor Classification and Signal Transduction. To browse other handbook pages, click here.
This compound was developed by Wyeth. To browse the list of other pharma-developed compounds and Approved Drugs/Drug Candidates, click here.
신호어
Warning
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
Qiong Xie et al.
Journal of molecular modeling, 12(4), 390-397 (2006-01-13)
Molecular docking has been performed to investigate the binding mode of (-)-meptazinol (MEP) with acetylcholinesterase (AChE) and to screen bis-meptazinol (bis-MEP) derivatives for preferable synthetic candidates virtually. A reliable and practical docking method for investigation of AChE ligands was established
A Schnabel et al.
Der Anaesthesist, 60(11), 995-1001 (2011-09-16)
Epidural regional analgesia is still recommended as the gold standard for obstetric analgesia due to its high efficacy and less depressing effects to the central nervous system. However, if absolute or relative contraindications for a regional anesthetic technique are present
Zhenqi Shi et al.
Life sciences, 77(20), 2574-2583 (2005-07-05)
The aim of this paper is to investigate the pharmacokinetic behavior of hydrochloride meptazinol (MEP) in plasma, cerebrospinal fluid (CSF) and cerebral cortex after intranasal administration (8 mg/kg) in male Sprague-Dawley rats. The pharmacokinetic study of intravenous administration (8 mg/kg)
C Rudolphi et al.
Journal of chromatography. B, Biomedical applications, 663(2), 315-326 (1995-01-20)
A reversed-phase high-performance liquid chromatographic method to separate meptazinol and its phase I metabolites has been developed using a LiChrosper 100 CN column and a mobile phase of trimethylammoniumacetate buffer (pH 5.5)-acetonitrile-methanol. Quantification of meptazinol and N-desmethylmeptazinol in biological samples
Wei Li et al.
Acta pharmacologica Sinica, 26(3), 334-338 (2005-02-18)
To investigate the mechanism of action of a potent analgesic, (+/-)-meptazinol. The structures of meptazinol enantiomers were compared with opioid pharmacophore and tramadol. Neither enantiomer of meptazinol fitted any patterns among the opioid pharmacophore and tramadol, although they did share
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.