Y0001510
Lactulose for peak identification
European Pharmacopoeia (EP) Reference Standard
동의어(들):
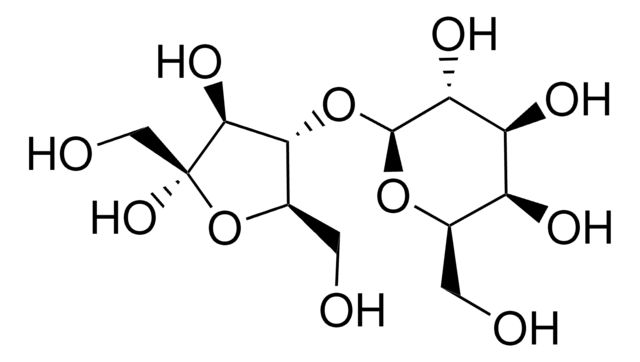
Lactulose, 4-O-β-D-Galactopyranosyl-D-fructofuranose, 4-O-β-D-Galactopyranosyl-D-fructose
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C12H22O11
CAS Number:
Molecular Weight:
342.30
Beilstein:
93773
MDL number:
UNSPSC 코드:
41116107
PubChem Substance ID:
NACRES:
NA.24
추천 제품
Grade
pharmaceutical primary standard
API family
lactulose
제조업체/상표
EDQM
응용 분야
pharmaceutical (small molecule)
형식
neat
저장 온도
2-8°C
SMILES string
OC[C@H]1O[C@@H](O[C@@H]2[C@@H](CO)O[C@@](O)(CO)[C@H]2O)[C@H](O)[C@@H](O)[C@H]1O
InChI
1S/C12H22O11/c13-1-4-6(16)7(17)8(18)11(21-4)22-9-5(2-14)23-12(20,3-15)10(9)19/h4-11,13-20H,1-3H2/t4-,5-,6+,7+,8-,9-,10+,11+,12+/m1/s1
InChI key
JCQLYHFGKNRPGE-WJONTELPSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
애플리케이션
Lactulose for peak identification EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
포장
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
기타 정보
Sales restrictions may apply.
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
Rifaximin for episodic, overt hepatic encephalopathy: the data are catching up to clinical practice, but questions remain.
Stephen E Congly et al.
The American journal of gastroenterology, 109(4), 598-598 (2014-04-05)
B Vogt et al.
Scandinavian journal of gastroenterology. Supplement, 222, 100-101 (1997-01-01)
The introduction of lactulose as a new therapeutic agent for treatment of hepatic encephalopathy was a major breakthrough in this field. It was hypothesized that lactulose might prevent postoperative renal impairment after biliary surgery in patients with obstructive jaundice. The
F L Weber
Digestive diseases (Basel, Switzerland), 14 Suppl 1, 53-63 (1996-01-01)
Lactulose is the most frequently utilized agent in the treatment of hepatic encephalopathy because of its efficacy and the fact that it has few serious side effects. How this nonabsorbable disaccharide works has been a matter of controversy, but evidence
Lactulose in clinical use.
J Ray et al.
Tropical gastroenterology : official journal of the Digestive Diseases Foundation, 17(2), 12-14 (1996-04-01)
S Salminen et al.
Scandinavian journal of gastroenterology. Supplement, 222, 45-48 (1997-01-01)
During the fermentation of lactulose, short-chain fatty acids are formed with consequent lowering of the colon pH and modification of the microflora. Lactulose promotes the growth of lactic acid bacteria and bifidobacteria and, more specifically, Lactobacillus acidophilus in the colon.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.