추천 제품
Grade
pharmaceutical primary standard
API family
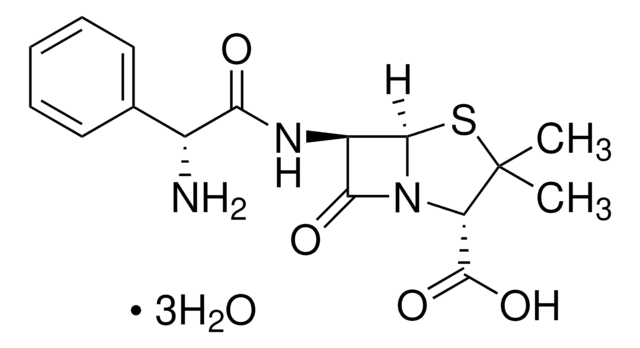
ampicillin
제조업체/상표
EDQM
mp
198-200 °C (dec.) (lit.)
응용 분야
pharmaceutical (small molecule)
형식
neat
저장 온도
2-8°C
SMILES string
O.O.O.CC1(C)S[C@@H]2[C@H](NC(=O)[C@H](N)c3ccccc3)C(=O)N2[C@H]1C(O)=O
InChI
1S/C16H19N3O4S.3H2O/c1-16(2)11(15(22)23)19-13(21)10(14(19)24-16)18-12(20)9(17)8-6-4-3-5-7-8;;;/h3-7,9-11,14H,17H2,1-2H3,(H,18,20)(H,22,23);3*1H2/t9-,10-,11+,14-;;;/m1.../s1
InChI key
RXDALBZNGVATNY-CWLIKTDRSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
애플리케이션
Ampicillin trihydrate EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
생화학적/생리학적 작용
A β-lactam antibiotic with an amino group side chain attached to the penicillin structure. Penicillin derivative that inhibits bacterial cell-wall synthesis (peptidoglycan cross-linking) by inactivating transpeptidases on the inner surface of the bacterial cell membrane. Bactericidal only to growing Escherichia coli . Mode of resistance: Cleavage of β-lactam ring of ampicillin by β-lactamase. Antimicrobial spectrum: Gram-negative and Gram-positive bacteria.
포장
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
기타 정보
Sales restrictions may apply.
관련 제품
제품 번호
설명
가격
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Resp. Sens. 1 - Skin Irrit. 2 - Skin Sens. 1 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
이미 열람한 고객
Felipe H Coutinho et al.
Microbial ecology, 68(3), 441-452 (2014-05-14)
Bacterial resistance to antibiotics has become a public health issue. Over the years, pathogenic organisms with resistance traits have been studied due to the threat they pose to human well-being. However, several studies raised awareness to the often disregarded importance
Hazem E Ghoneim et al.
The Journal of infectious diseases, 209(9), 1459-1468 (2013-11-26)
Secondary bacterial pneumonia is a significant cause of morbidity and mortality during influenza, despite routine use of standard antibiotics. Antibiotic-induced immunopathology associated with bacterial cell wall lysis has been suggested to contribute to these poor outcomes. Using Streptococcus pneumoniae in
Yuraporn Sahasakul et al.
The British journal of nutrition, 112(3), 408-415 (2014-06-17)
Lactobacilli are indigenous microbes of the stomach of rodents, with much lower numbers being present in mice fed a purified diet than in those fed a non-purified diet. We postulated that gastric emptying (GE) is responsible for the different colonisation
Bacterial endocarditis following lithotripsy: an unusual complication caused by a non-invasive procedure.
Alessandra Soriano et al.
The Israel Medical Association journal : IMAJ, 16(12), 803-804 (2015-01-30)
Ebtehal S Al-Abdullah et al.
International journal of molecular sciences, 15(12), 22995-23010 (2014-12-17)
The reaction of 5-(1-adamantyl)-4-ethyl or allyl-1,2,4-triazoline-3-thione with formaldehyde solution and various 1-substituted piperazines yielded the corresponding N-Mannich bases. The newly synthesized N-Mannich bases were tested for in vitro inhibitory activities against a panel of Gram-positive and Gram-negative bacteria and the
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.