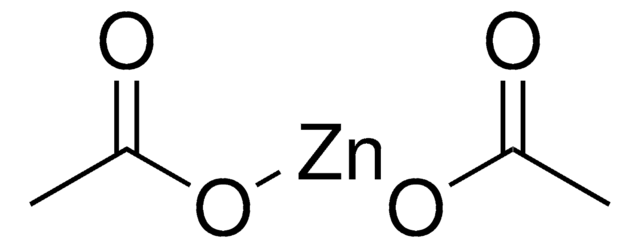
추천 제품
Grade
ACS reagent
Quality Level
vapor pressure
1 mmHg ( 487 °C)
분석
≥99.8%
양식
granular
반응 적합성
core: zinc
reagent type: catalyst
저항도
5.8 μΩ-cm, 20°C
입자 크기
20-30 mesh
bp
907 °C (lit.)
mp
420 °C (lit.)
density
7.133 g/mL at 25 °C (lit.)
양이온 미량물
Fe: ≤0.01%
Pb: ≤0.01%
적합성
passes application test for determination of arsenic (As)
SMILES string
[Zn]
InChI
1S/Zn
InChI key
HCHKCACWOHOZIP-UHFFFAOYSA-N
일반 설명
Zinc granules are pure metallic zinc in a granular form, they provide a high surface area for increased reactivity. They serve as a reducing agent, catalyst, or precursor for various chemical reactions. Zinc granules are commonly used for synthesis of organometallic compounds. It reacts with organic halides to form alkyl or aryl zinc compounds, which serve as powerful nucleophiles in carbon-carbon bond formations. This reaction, known as the Barbier reaction, enables the introduction of diverse functional groups into organic molecules.Zinc granules also find application in the synthesis of pharmaceuticals and fine chemicals. They can be employed in the reduction of functional groups like nitro, nitrile, or azide to their corresponding amines. Additionally, zinc granules are involved in other carbon-carbon bond forming reactions, such as the Reformatsky reaction and the Simmons-Smith reaction.
애플리케이션
Zinc granules can be used to prepare:
- organozinc reagents commonly used in Negishi or Fukuyama couplings
- Reformatsky reagents
- Simmons-Smith reagents
신호어
Warning
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
이미 열람한 고객
Use of activation methods for organozinc reagents.
Erdik E
Tetrahedron, 43(10), 2203-2212 (1987)
A Convenient Procedure for the Preparation of Reactive Zinc for the Reformatsky Reaction.
Bouhel E
Synthetic Communications, 21(1), 133-136 (1991)
A scalable zinc activation procedure using dibal-h in a reformatsky reaction.
Girgis M J
Organic Process Research & Development, 13(6), 1094-1099 (2009)
Cyclopropanation of Electron-deficient Olefins with Dibromomethane by Ni (0) Complexes and Zinc.
Kanai H
Bulletin of the Chemical Society of Japan, 56(4), 1025-1029 (1983)
Cyclopropanes from an Easily Prepared, Highly Active Zinc?Copper Couple, Dibromomethane, and Olefins.
LeGoff E
The Journal of Organic Chemistry, 29(7), 2048-2050 (1964)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.