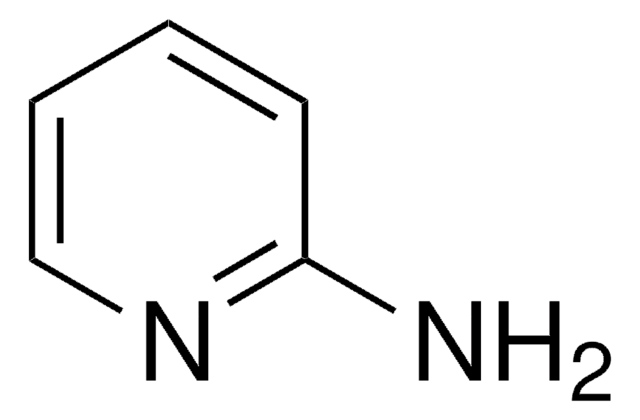
추천 제품
Quality Level
분석
95%
양식
solid
refractive index
n20/D 1.5823 (lit.)
bp
221-222 °C (lit.)
mp
29-31 °C (lit.)
density
1.073 g/mL at 25 °C (lit.)
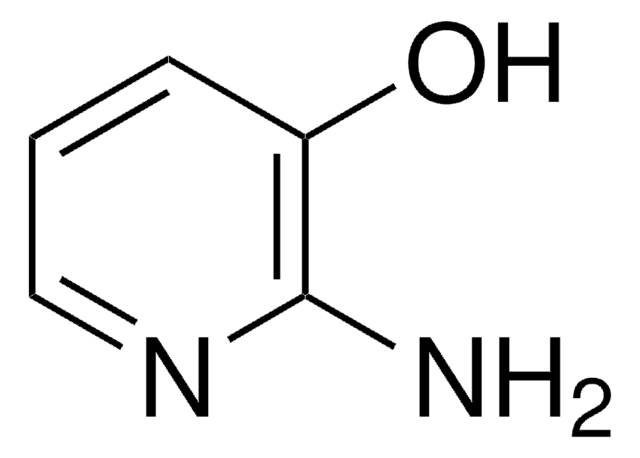
SMILES string
Cc1cccnc1N
InChI
1S/C6H8N2/c1-5-3-2-4-8-6(5)7/h2-4H,1H3,(H2,7,8)
InChI key
RGDQRXPEZUNWHX-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 2 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Chronic 3 - Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 2
Flash Point (°F)
244.4 °F - closed cup
Flash Point (°C)
118 °C - closed cup
개인 보호 장비
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
이미 열람한 고객
T G Altuntas et al.
Anticancer research, 17(5A), 3485-3491 (1997-12-31)
The metabolism of the comutagen 2-amino-3-methylpyridine has been studied in vitro using rat and rabbit hepatic preparations. 2-Amino-3-methylpyridine-N-oxide, 2-amino-3-hydroxymethylpyridine and 2-amino-5-hydroxy-3-methylpyridine were formed by both rat and rabbit hepatic preparations. No evidence was obtained for the formation of the corresponding
J W Gorrod et al.
European journal of drug metabolism and pharmacokinetics, 22(4), 341-344 (1998-03-26)
The in vitro metabolism of 2-hydroxylamino-3-methylpyridine has been investigated using arochlor 1254 pretreated rat S9 mixtures. 2-Hydroxylamino-3-methylpyridine is rapidly converted to the parent amine 2-amino-3-methylpyridine. No further oxidation products of 2-hydroxylamino-3-methylpyridine (i.e. nitroso or nitro) were detected under the HPLC
T G Altuntas et al.
Anticancer research, 17(6D), 4479-4482 (1998-03-12)
The metabolism of 2-amino-3-methylpyridine (2A3MP) in vitro has been investigated using the rat, rabbit, dog, marmoset, guinea pig and hamster hepatic microsomes and S9 supernatants (10,000 g fraction). Species differences were observed in the in vitro formation of 2-amino-3-methylpyridine-N-oxide (2A3MPNO)
Cengiz Yenikaya et al.
Journal of enzyme inhibition and medicinal chemistry, 26(1), 104-114 (2010-09-24)
Two novel proton transfer compounds were prepared between 2,4-dichloro-5-sulphamoylbenzoic acid (lasamide) (Hsba) and ethylenediamine (en), namely ethane-1,2-diaminium 2,4-dichloro-5-sulphamoylbenzoate (1), and also between Hsba and 2-amino-3-methylpyridine (2-amino-3-picoline) (amp), namely 2-amino-3-methylpyridinium 2,4-dichloro-5-sulphamoylbenzoate (2). All these were characterised by elemental, spectral (IR and
T G Altuntas et al.
Xenobiotica; the fate of foreign compounds in biological systems, 27(11), 1103-1111 (1997-12-31)
1. The effects of various potential inhibitors, activators and inducers on the metabolism of the comutagen 2-amino-3-methylpyridine (2A3MP) by rabbit hepatic microsomes and S9 supernatants have been studied. 2. The 1-N-oxidation of 2A3MP to 2-amino-3-methylpyridine-1-N-oxide (2A3M-PNO) was inhibited by 2,4-dichloro-6-phenylphenoxyethylamine
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.