105171
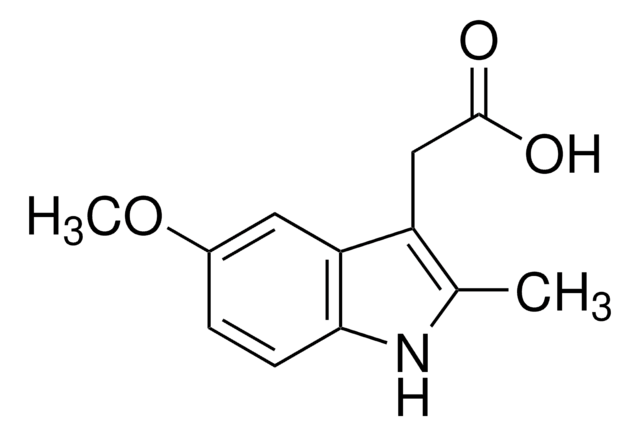
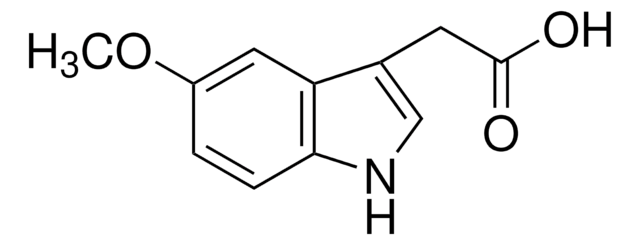
5-Methoxy-2-methyl-3-indoleacetic acid
98%
동의어(들):
N-Des(4-chlorobenzoyl)indomethacin, NSC 97026, 2-(5-Methoxy-2-methyl-1H-indol-3-yl)acetic acid
로그인조직 및 계약 가격 보기
모든 사진(3)
About This Item
실험식(Hill 표기법):
C12H13NO3
CAS Number:
Molecular Weight:
219.24
EC Number:
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
98%
mp
161-163 °C (lit.)
solubility
methanol: soluble
작용기
carboxylic acid
SMILES string
COc1ccc2[nH]c(C)c(CC(O)=O)c2c1
InChI
1S/C12H13NO3/c1-7-9(6-12(14)15)10-5-8(16-2)3-4-11(10)13-7/h3-5,13H,6H2,1-2H3,(H,14,15)
InChI key
TXWGINUZLBAKDF-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
관련 카테고리
일반 설명
5-Methoxy-2-methyl-3-indoleacetic acid is hydrolysis product of indomethacin.
애플리케이션
5-Methoxy-2-methyl-3-indoleacetic acid was used for quantitative determination of indomethacin and its major impurities in suppository and capsule formulations by HPLC. 5-Methoxy-2-methyl-3-indoleacetic acid was used in a study to develop fast, sensitive and simultaneous determination of metabolites of serotonin using liquid chromatography with mass spectrometric detection.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
W I Rosenblum et al.
Prostaglandins, 21(4), 667-673 (1981-04-01)
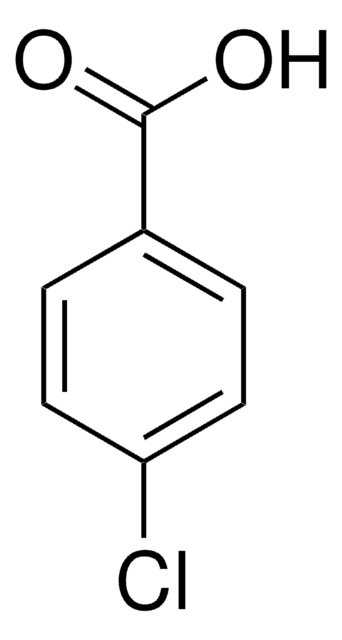
Previous studies suggested that one of the hydrolysis products of indomethacin, either 4-chlorobenzoic acid or 5-methoxy-2-methyl-3-indole acetic acid, can inhibit platelet aggregation in vivo. If correct, this hypothesis explains the apparent action of indomethacin dissolved at high pH where hydrolysis
T B Vree et al.
Journal of chromatography, 616(2), 271-282 (1993-07-02)
Indomethacin is metabolized in humans by O-demethylation, and by acyl glucuronidation to the 1-O-glucuronide. Indomethacin, its metabolite O-desmethylindomethacin (DMI) and their conjugates can be measured directly by gradient high-performance liquid chromatographic analysis without enzymic deglucuronidation. The glucuronide conjugates were isolated
H Alho et al.
Cell growth & differentiation : the molecular biology journal of the American Association for Cancer Research, 5(9), 1005-1014 (1994-09-01)
A recognition site for benzodiazepines structurally different from that linked to the gamma-aminobutyric acid receptor subtype A or the "central type" benzodiazepine receptor has been located mainly in the outer membranes of mitochondria and designated mitochondrial benzodiazepine receptor (MBR). A
Peng Zhang et al.
Acta pharmacologica Sinica, 27(8), 1097-1102 (2006-07-27)
To investigate the biotransformation of indomethacin, the first of the newer nonsteroidal anti-inflammatory drugs, by filamentous fungus and to compare the similarities between microbial transformation and mammalian metabolism of indomethacin. Five strains of Cunninghamella (C elegans AS 3.156, C elegans
E Kwong et al.
Journal of pharmaceutical sciences, 71(7), 828-830 (1982-07-01)
Indomethacin and its impurities in suppository and capsule formulations were quantitatively determined by HPLC using a reversed-phase, octadecyl column and a mobile phase of methanol-water-acetonitrile-acetic acid (55:35:10:1). Analysis of the suppository formulations provided a mean potency for indomethacin of 103.8%.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.